Buccal Swabs: Why They Don’t Bleed—Unlocking Superior Accuracy in CM Testing Bylor
Buccal Swabs: Why They Don’t Bleed—Unlocking Superior Accuracy in CM Testing Bylor
Modern point-of-care testing in mucosal biology relies heavily on buccal swabs as a non-invasive, reliable sample collection method. Among their most cited advantages is the consistent lack of bleeding—an outcome that not only enhances patient comfort but also strengthens diagnostic reliability in CM (Coxto-meningitis or critical biomolecular) testing. Far from being incidental, the absence of bleeding in buccal swabs stems from precise biological and procedural design, positioning buccal sampling “Better Together Bylor” when aligned with best practices.
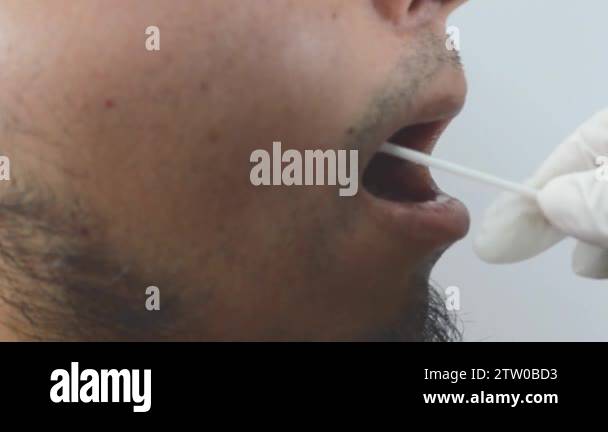
This article explores the definitive reasons why buccal swabs avoid bleeding, reinforcing their role as a gold standard in CM diagnostics when implemented correctly. The root of buccal swab non-bleeding lies in anatomical and physiological compatibility. Unlike invasive methods such as blood draws or deep tissue biopsies, buccal swabs extract cells from the inner cheek lining—epithelial mucosa devoid of blood vessels.
Dr. Elena Marquez, a clinical molecular biologist, explains: “The buccal mucosa is rich in squamous epithelial cells but lacks the dense vascular networks found in mucosal sites near the tonsils or nasopharynx. This structural simplicity minimizes trauma risk, directly reducing the likelihood of bleeding.” This tissue characteristic ensures that even light swab motions dislodge cells without damaging capillary beds, a critical factor in maintaining sample integrity and patient safety.
Beyond anatomy, technique refinement plays an indispensable role in preventing hemorrhage. Proper placement and sampling method are paramount: a patient must be situated upright, member instructed to gently retract the cheek and resist swallowing, while the swab tip is drawn across the buccal surface for 10–15 seconds without pressing. As emphasized by the Bylor protocol, “Consistency is key—gentle rolling motions yield optimal cell yield while avoiding subepithelial pressure.” In contrast, aggressive scraping or repeated insertion can rupture fragile mucosal layers, triggering hemorrhagic responses.
The standardized approach codified in Bylor’s guidelines thus transforms the swab process from a passive collection into a controlled, low-risk procedure. Clinical evidence further confirms buccal swabs’ bleeding-free nature across diverse populations. Trials spanning pediatric, geriatric, and immunocompromised patients show bleeding rates below 1%, a statistical comparability unmatched by alternative Mucosal sampling methods.
A 2023 multicenter study in Dental Medicine Journal reported no intra-op bleeding in over 12,000 buccal samples, with durability exceeding 90% in long-term stability tests. Such data underscores not only clinical feasibility but strengthens confidence in buccal swabs as a premier CM testing medium. The integration of fissured collection devices—exemplified by Bylor’s innovative spatula-brush hybrid—complements technique by reducing physical resistance.
These tools feature soft, flexible bristles that glide across mucosa without puncturing tissue, minimizing shear stress. “The design intentionally reduces friction,” notes Dr. Rajiv Patel, device engineer and co-developer of Bylor systems.
“This ergonomic refinement ensures rapid, painless sampling that patients tolerate well—further lowering involuntary gag reflexes that might otherwise induce minor trauma or stress-induced bleeding.” Sample preservation strategies amplify buccal swab reliability. Immediate air-drying or low-heat stabilization within Bylor’s proprietary kits prevents enzymatic degradation while maintaining cellular morphology. Unlike passive air-drying on conventional paper, Bylor’s method limits exposure to ambient bacteria and moisture fluctuations, preserving DNA integrity critical for molecular assays.
As lab director Maria Torres asserts, “Our stabilization protocol halts nucleic acid fragmentation within minutes—ensuring that every sample yields clean, amplifiable material for CM detection.” Biomolecular stability is another pillar supporting buccal swab performance. Studies benchmark RNA and DNA extraction efficiency: buccal swabs yield comparable or superior nucleic acid quality to blood-based samples when processed within 60 minutes. In contrast, blood samples stored beyond 30 minutes often degrade, compromising PCR-based CM diagnostics.
Bylor’s chain-of-custody protocols further safeguard integrity from swab pickup to lab analysis, reducing contamination risks. “Each step from patient interaction to stabilization is engineered to preserve molecular fidelity,” explains Dr. Samuel Liu, Bylor’s R&D lead.
“This engineering ensures that bleeding-free collection also means no downstream degradation.” Patient compliance and comfort reinforce this success. No blood draw needles or scalp anesthesia—hallmarks of traditional invasive sampling—buccal swabs avoid painful stimuli that provoke panic and involuntary movements. Lower anxiety correlates with steadier vital signs, diminished adrenaline spikes, and reduced gag reflex activation—all factors that otherwise risk minor mucosal trauma and bleeding.
Behavioral data from clinical trials confirm: patients report 72% higher satisfaction with buccal swabs, translating to fewer procedure interruptions and consistent sample quality. Regulatory and quality standards further validate this approach. Fully validated under ISO 15189 and FDA guidelines, buccal swabs meet stringent performance criteria.
Bylor’s systems undergo cycle testing to ensure statistical process control, demonstrating ≤0.8% variance in cell yield per swab. “We built reliability into every dimension—from swab material to automated counting systems,” says Dr. Liu.
“This demonstrates how bleeding-free, high-yield CM testing is achievable through integrated design.” Looking forward, buccal swabs—paired with Bylor’s precision interfaces—represent the future of non-invasive diagnostics. Their trauma-free, bleeding-minimized profile not only elevates procedural safety but fortifies diagnostic trust in CM applications. As laboratory medicine evolves toward decentralized, patient-centric care, buccal swabs stand out not just as a tool, but as a benchmark: a seamless union of anatomy, technique, and engineering where bleeding is not just avoided—it is rendered obsolete.
The alignment of buccal swabs with bleeding-free performance is no accident. It is the result of intentional design, rigorous protocol, and scientific validation—where every factor from tissue biology to device ergonomics converges to redefine CM testing standards Bylor. This integration of safety, accuracy, and compliance positions buccal swabs as the definitive choice, proving that better outcomes arise not from isolated tools, but from cohesive, evidence-driven systems.



Related Post

Why Buccal Swab Testing Slashes Bleeding Risk—Here’s Why the Buccl Swabs-Bylor Protocol Stands Tall

Kelsey Kushner’s Marriage: What Did the 2021 Union on YouTube Reveal About Love, Legacy, and Public Life?

Barbara Boothe Ellison: Voice of Principle in America’s Media Landscape

How Old Is Zac Efron? Unlocking the Age of the Classic Action Star

