Decoding the Oxygen Atom: How Lewis Dot Structure Reveals the Heart of Chemical Reactivity
Decoding the Oxygen Atom: How Lewis Dot Structure Reveals the Heart of Chemical Reactivity
Oxygen, the most abundant element in Earth’s atmosphere and a cornerstone of life, holds profound influence over chemical behavior—determining how molecules form, react, and sustain biological processes. At the core of understanding oxygen’s role lies a deceptively simple yet powerful tool: the Lewis dot structure. By mapping the valence electrons and predicting molecular connectivity, this foundational model exposes the subtle dynamics that drive reactivity.
Far more than a static image, the Lewis structure decodes the electron-sharing strategies that define oxygen’s chemical personality.
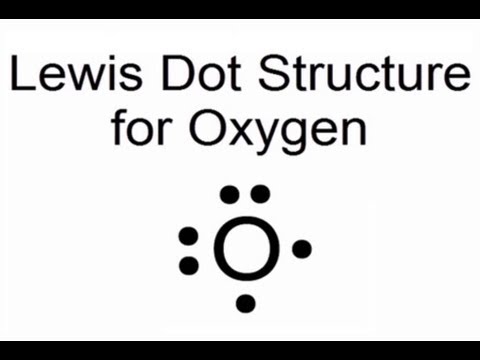
Central to this visualization is the concept of valence electrons—electrons in an atom’s outermost energy level available for bonding. Oxygen, with six valence electrons (as illustrated in Group 16 of the periodic table), seeks stability through electron pair sharing or transfer.
The Lewis dot structure clearly shows oxygen bearing two unpaired electrons in its p orbital, ready to form bonds and stabilize its electronic configuration. Each dot represents a physical electron, but their arrangement reveals deeper reactivity: oxygen rarely acts alone, preferring to form diatomic molecules (O₂), water (H₂O), or oxides (e.g., SiO₂), where its electron interactions define chemical outcomes.
Electron Arrangement: Beyond the Surface
Oxygen’s electron configuration—1s² 2s² 2p⁴—dictates its chemical nature.The 2p orbitals are critical: they contain four electrons capable of forming multiple bonds or lone pairs, enabling oxygen to engage in hydrogen bonding, coordinate with metals, and support redox reactions. In Lewis theory, bonding arises from shared pairs, while lone pairs influence molecular geometry and reactivity. A key insight is that oxygen’s preference for two bonds (common in compounds like water) stems from its desire to achieve an octet, a thermodynamic drive for electron stability.
This shapes how oxygen interacts with hydrogen, carbon, and transition metals.
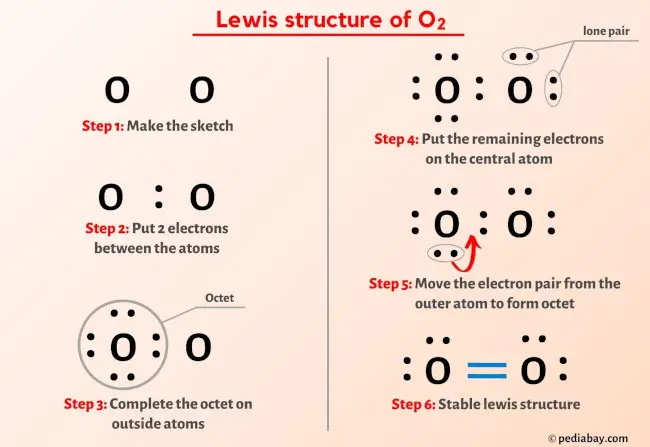
Consider the oxygen atom in O₂: its two unpaired electrons in separate p orbitals explain the molecule’s paramagnetic behavior and high bond strength. Dividing the electron count, each oxygen contributes two unpaired spins—stable yet reactive under certain conditions, enabling combustion and respiration.
In water (H₂O), oxygen forms two covalent bonds with hydrogen, leaving two lone pairs that bend the molecule’s shape via VSEPR theory. This bended geometry increases polarity, enhancing water’s solvent power and hydrogen bonding capacity—critical for life’s hydration processes.
From Thomas Lewis to Modern Chemistry
The modern visualization of atomic bonding traces back to Gilbert N.Lewis, whose 1916 representation of electron pairs as dots around atoms revolutionized chemical thinking. Though a simplification, Lewis structures vividly communicate valence electron dynamics, capturing: - bonding pairs: shared between atoms - lone pairs: non-bonding electrons contributing to molecular shape - polyatomic ions: such as CO₃²⁻, where oxygen’s charge distribution is inferred Marie L. Flynn, a chemistry education researcher, notes, “Lewis structures are not perfect, but they bridge intuition and quantum mechanics—making complex reactivity accessible.” These diagrams reveal how oxygen’s electron tap—seeking closure through bonding—underpins critical biological and industrial reactions.
Oxygen’s Role Across Reactive Scenarios
In combustion, oxygen’s electron demand drives explosive reactions: hydrocarbons burn because oxygen electrons accept free electrons, forming stable O–H and O–C bonds while releasing energy. In biological systems, hemoglobin’s iron binds O₂ reversibly, exploiting oxygen’s willingness to form transient coordination complexes. In oxides like aluminum oxide (Al₂O₃), oxygen’s hybridization supports network covalent structures, conferring high melting points and corrosion resistance.The Lewis structure captures these varied behaviors through apparent electron count: whether showing full octets in stable molecules or transient lone pairs during redox processes, it translates electron behavior into molecular function.
Implications for Material Science and Drug Design
Beyond fundamental chemistry, understanding oxygen’s lewire dynamics informs material innovation and pharmaceutical development. Catalysts in catalytic converters rely on oxygen’s reactivity—promoting selective oxidation of pollutants.In drug design, oxygen’s hydrogen-bonding capacity guides target binding; antioxidants neutralize free radicals by donating electrons, stabilizing oxygen-containing reactive species. “Structural clarity from Lewis representations accelerates discovery,” says Dr. Elena Petrova, a computational chemist.
“We design molecules with precision by knowing exactly how oxygen electron clouds interact—whether forming weak dipoles or strong bonds.”
This predictive power underscores why decoding the oxygen atom through Lewis dot structure remains essential. It transforms abstract quantum behavior into tangible chemical insight—revealing the silent moves that sustain life, power industry, and inspire innovation. From lab to life, oxygen’s chemistry begins with a dot, but its impact resonates across the spectrum of chemical reality.


Related Post

Robert Hawking: The Genius Who Mapped Black Holes, Challenged Gravity, and Redefined Cosmology
K3 Visa Processing Time 2025: Plan Your Journey with Precision

Ashlee Kyle Palmieri's Role In Kyle Palmieri's Success

Lieserl The Enigmatic Daughter Of Albert Einstein

