Electron vs. Molecular Geometry: Decoding Two Worlds That Shape Technology and Chemistry
Electron vs. Molecular Geometry: Decoding Two Worlds That Shape Technology and Chemistry
In the ever-evolving landscape of modern technology and molecular science, two seemingly disparate domains—Electron behavior in soft-matter systems and rigid molecular geometry—converge in their fundamental influence on innovation and understanding. Electron Vs Molecular Geometry explores the contrasting yet complementary realms where subatomic particles and atomic frameworks define everything from computational performance to chemical reactivity. While one operates in the fluid, dynamic domain of quantum mechanics and electronic motion, the other governs the precise spatial arrangements that determine molecular function.
Understanding their distinctions and interplay offers insight into breakthroughs in materials science, pharmaceuticals, and next-generation computing.
Electrons, the fundamental constituents of matter, govern near-instantaneous interactions in physical and digital systems. Their quantum nature—exhibiting wave-particle duality and probabilistic distribution—underpins the design of nanoelectronic devices and energy transfer mechanisms.
In contrast, molecular geometry refers to the three-dimensional arrangement of atoms within a molecule, dictated by electron pair repulsion and bond formation. This spatial configuration dictates chemical stability, reactivity, and biological activity, making it essential in drug development and catalyst design. The juxtaposition reveals a deeper narrative: how atomic-scale precision and electron dynamics jointly enable advancements across fields from electronics to medicine.
Unpacking Electron Vs Molecular Geometry: Key Contrasts and Concepts
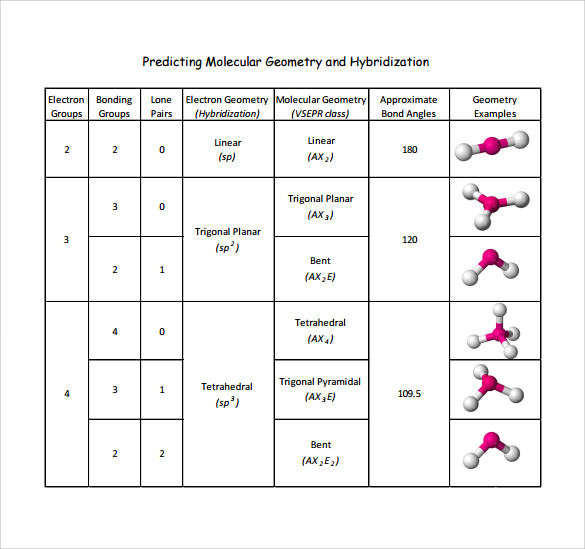
At the core of the divide between Electron Vs Molecular Geometry lies a fundamental difference in scale, behavior, and application.Electrons exist at the quantum level, where their positions are defined by probability clouds rather than fixed locations, and their interactions can span nanoseconds or less in high-speed devices. Molecular geometry, by contrast, is rigorously deterministic—described by well-established rules like Valence Shell Electron Pair Repulsion (VSEPR) and hybridization theory, dictating molecular shape with high predictability. *Electrons: Dynamic and Ephemeral* Electrons move rapidly through space and time, driven by electromagnetic forces.
Their energy states and orbital distributions are described by Schrödinger’s equation and probabilistic models. This fluidity enables phenomena such as tunneling in semiconductors and charge transfer in batteries. “Electron behavior is probabilistic and fast—orders of magnitude quicker than molecular conformational changes,” explains Dr.
Helen Moss, quantum chemist at Stanford University. “This dynamism enables devices that operate at terahertz frequencies but complicates their deterministic modeling.” *Molecules: Structural Precision Meets Stability* Molecular geometry refers to the static or quasi-static arrangement of atoms within a molecule, governed by covalent bonding, steric effects, and electron repulsion. The VSEPR model, for example, predicts shapes—linear, trigonal planar, tetrahedral—by minimizing repulsion between bonding and lone pairs.
Such structural predictability allows chemists to anticipate molecular behavior, design catalysts, and develop pharmaceuticals with targeted activity. The precision of molecular geometry is why a single atom’s orientation within a drug molecule can determine its therapeutic success or failure.
Applications Where Electron Dynamics and Molecular Structure Converge
The divergence between Electron Vs Molecular Geometry is not just theoretical—it drives real-world innovation across multiple industries.In drug discovery, molecular geometry determines how a compound binds to a target protein, but electron distribution governs binding affinity and signal transduction. Similarly, in semiconductor technology, electron mobility dictates device speed, while the geometry of atomic lattices influences material conductivity and thermal stability. Consider aerospace materials: lightweight composites derive strength from molecular alignment and electron delocalization in polymer chains.
In lithium-ion batteries, electron transfer across electrode surfaces depends not only on molecular interface design but also on electronic conductivity patterns shaped by atomic arrangement. “Electrons move fast, but they require a molecular highway to follow,” notes Dr. Raj Patel, materials engineer at MIT.
“Optimizing both layers unlocks breakthroughs in efficiency and durability.”
Beyond chemistry and electronics, the interaction between electron behavior and molecular geometry is foundational in nanotechnology, photovoltaics, and even biological signaling. In perovskite solar cells, for instance, electron transport and molecular stacking determine energy conversion efficiency. Likewise, enzyme catalysis relies on precisely positioned atoms to orient substrates while facilitating rapid electron transfer.
These examples illustrate how molecular architecture enables electron mobility, and vice versa, forming a feedback loop that propels scientific progress.
The Quantum Constraint: Electrons as Architects of Molecular Form
Though distinct in behavior, electrical dynamics indirectly shape molecular geometry through quantum mechanical constraints. Electron-electron repulsion pushes atoms into specific spatial configurations, making VSEPR and hybridization theory not just descriptive tools but predictive frameworks rooted in physical laws. In transition metal complexes, d-orbital interactions modulate both electron character and spatial symmetry, linking electron configuration directly to molecular shape.“Molecular geometry emerges from electron behavior at its most intimate scale,” explains Professor Elena Volkov, quantum structure theorist at ETH Zurich. “Electrons aren’t just passive players—they design the stage upon which geometry unfolds.”
Challenges in Modeling Both Domains
Accurately simulating Electron Vs Molecular Geometry demands distinct computational approaches, each with limitations. Electron behavior requires advanced quantum chemical methods—like density functional theory (DFT) or time-dependent DFT—that balance accuracy with computational cost, especially for large systems.These methods approximate electron correlation but struggle with strongly correlated materials or excited states. Molecular geometry, while more tractable, involves combinatorial complexity in predicting optimal conformations across large molecule spaces. Force fields and molecular dynamics simulations offer reasonable approximations but may overlook subtle electronic effects.
Hybrid quantum mechanics/molecular mechanics (QM/MM) methods attempt to bridge this gap, modeling electron activity in active sites while treating the rest of the system classically. Yet full integration remains an open frontier. “No single framework captures all nuances,” cautions Dr.
Volkov. “Electron dynamics demand quantum fidelity, while molecular geometry rewards efficient spatial prediction—two worlds requiring separate but complementary tools.”
Future Horizons: Synergizing Electron Control with Molecular Design
The convergence of Electron Vs Molecular Geometry heralds a new era in materials innovation. Advances in machine learning and artificial intelligence now enable faster simulations, predicting both electron behavior and molecular stability with unprecedented speed.Quantum computing promises to resolve longstanding limitations in electron correlation modeling, unlocking deeper insights into catalysis and superconductivity. In pharmaceuticals, “precision medicine hinges on aligning drug molecule geometry with targeted electronic interactions—an integration of form and function,” asserts Dr. Moss.
Meanwhile, next-gen semiconductors exploit tailored electron pathways within atomically precise molecular frameworks, pushing computing performance toward theoretical limits. Ultimately, Electron Vs Molecular Geometry is not a competition but a collaboration—one where quantum motion meets spatial design to shape technologies that transform daily life. As science learns to navigate both domains with finer precision, the boundary between what electrons do and what molecules are becomes increasingly porous, revealing deeper synergies that drive discovery forward.
From tiny drug molecules to vast nanoengineered systems, the interplay between electron behavior and molecular architecture remains the silent architect of innovation—quiet, powerful, and profoundly essential.



Related Post

Inside General Hospital Star Kelly Monac: Shaping Melbourne’s Medical Future One Story at a Time

Rachael Speed’s Path to Redefining Resilience in Mental Health Advocacy

Unveiling The Personal Life Of Gareth Bale: Who Is The Man Behind The Goals and the Private Seasons

Jacqueline Innes Wood: Architectural Visionary Shaping Modern Memory

