Is Solubility a Physical or Chemical Property? Unlocking the Science of How Substances Dissolve
Is Solubility a Physical or Chemical Property? Unlocking the Science of How Substances Dissolve
Everyday choices—from dissolving sugar in coffee to understanding drug delivery systems—depend on a fundamental principle of chemistry: solubility. But is solubility merely a physical trait, or does it stem from deeper chemical transformations?
This article explores the nuanced nature of solubility, revealing how dissolution operates at both physical and chemical levels, governed by molecular interactions and energy dynamics. Far more than a simple mixing process, solubility shapes everything from environmental transport to pharmaceutical efficacy, relying on complex forces that determine what dissolves—and how quickly.
The Dual Nature of Solubility: Physical or Chemical?
At first glance, solubility appears purely physical—a measure of how many solute particles disperse in a solvent. Yet a closer examination reveals layers of chemical involvement that complicate this classification.
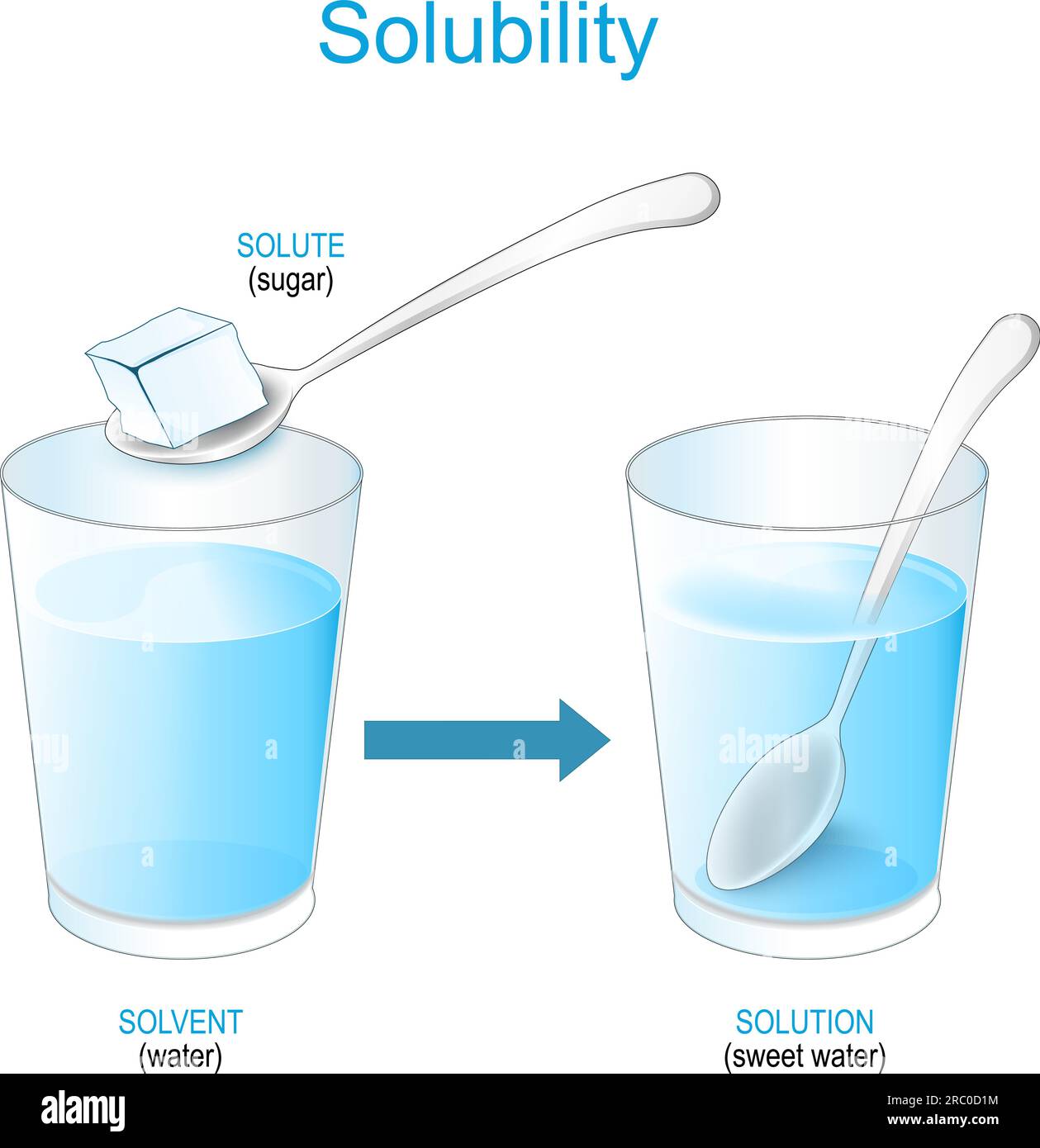
Solubility, defined as the maximum amount of solute that dissolves in a given quantity of solvent at equilibrium, is often taught as a physical property tied to temperature, pressure, and molecular polarity. However, when dissolution triggers chemical changes—such as ionization or bond breaking—solubility crosses into the realm of chemistry. “Solubility isn’t just about how easily a substance mixes; it’s about whether dissolution fundamentally alters the molecular identity of the solute,” notes Dr.
Elena Ramirez, a physical chemist at the Institute for Molecular Sciences.
Physical Aspects: The Classical View
From a physical sciences perspective, solubility aligns with classic behaviors observed in phase equilibria. Dissolution involves breaking intermolecular forces—like hydrogen bonds or dipole interactions—between solute particles and solvent molecules without changing their internal structure. “When table sugar (sucrose) dissolves in water, individual molecules separate and integrate into the solvent without breaking covalent bonds,” explains professor Marcus Lin, external expert in chemical thermodynamics.
This process is entropically driven: disorder increases as solute particles spread throughout the solvent, favoring dissolution in line with the Second Law of Thermodynamics.
- Solubility is temperature-dependent: most solids become more soluble as temperature rises, though gases typically show inverse behavior.
- Pressure affects only gases; solubility of solids and liquids increases slightly with pressure, per Henry’s Law.
- Polar solvents like water dissolve polar solutes—“like dissolves like”—based on matching intermolecular forces.
- The physical process is reversible and leaves the solute chemically unchanged—no reactions occur.
Chemical Mechanisms: Beyond Disintegration
Where solubility involves more than mere mixing, dissolution triggers chemical transformations, blurring the line between physical and chemical properties. When ionic compounds dissolve in water, ion-dipole interactions break lattice bonds, releasing individual ions into solution—processes with clear chemical implications.
“For salts like sodium chloride, dissolution involves endothermic bond breaking in the crystal and exothermic ion-solvent energy release,” says Dr. Ramirez.
“The free energy change determines spontaneity—dissolution happens not just because it’s favorable, but because the system’s chemistry favors new equilibria.”
In acid-base chemistry, solubility often reflects protonation states and pH-dependent dissolution—chemical reactions altering solute identity. Calcium phosphate, for example, is sparingly soluble but dissolves in acidic conditions when protons enhance solubility through protonation, forming soluble complexes. Similarly, certain metals—and even some organic dyes—dissolve only after undergoing redox or ligand-exchange reactions, underscoring how solubility can be inseparable from chemical change.
- Dissolution of ionic compounds (e.g., NaCl) involves breaking ionic bonds and hydrating ions—a chemical process.
- Gas solubility in water often includes weak chemical reactions (e.g., CO₂ forming carbonic acid), merging physical dissolution with chemical reaction.
- Polymer solubility may depend on chain unfolding or chemical compatibility, not just dispersion.
- pH-sensitive solutes exhibit solubility changes due to chemical equilibria involving protonation/deprotonation.
Practical Implications: From Nature to Industry
Understanding solubility’s dual character is not merely academic—it drives breakthroughs across science and technology.
In environmental science, the solubility of pollutants determines bioavailability: mercury’s weak solubility limits toxicity, while nitrate salts dissolve readily, contaminating groundwater. In medicine, drug solubility governs absorption in the human body—poorly soluble drugs challenge pharmaceutical design, prompting innovations like nanoformulations or co-solvents.
Agriculture leverages solubility for fertilizer efficiency; phosphorus availability depends on soil pH and mineral solubility, influencing crop yields. In industrial chemistry, controlling solubility enables precise reactions—from crystallization processes to carbon capture using solvents engineered to selectively dissolve specific gases.
---Solubility, therefore, is neither purely physical nor purely chemical, but a dynamic intersection where molecular forces and energy dictate behavior.
Whether viewed through thermodynamic equilibrium or reaction pathways, this process remains central to understanding how substances interact in mixtures. As science advances, recognizing solubility’s complexity empowers better predictions—transforming everyday mysteries into tools for innovation.
The science behind dissolution reveals a subtle but profound truth: solubility reflects a dance between dispersion and transformation, where physical movement and chemical affinity converge to shape what dissolves—and how it reshapes the world at microscopic and macroscopic scales.



Related Post

Is Solubility a Physical or Chemical Property? Unlocking the Science Behind Dissolution

Qball Shark Tank Founder: How One Innovator Built a Shorefront Empire and Shaped a Billion-Dollar Investment Story

Harold From The Five: Architect of Narrative Truth in a World of Fragmented Storytelling

Summer Tiger Age: Unlocking the Vital Window of Opportunity for Young Minds

