Mastering Chemical Clarity: Deciphering Lewis Structures with the HBR Approach
Mastering Chemical Clarity: Deciphering Lewis Structures with the HBR Approach
Understanding molecular architecture through Lewis structures is essential for chemists, educators, and students alike—these simple yet powerful diagrams reveal the invisible dance of electrons that govern chemical behavior. The Harvard Business Review (HBR) has highlighted how visual clarity in complex systems underpins strategic thinking and precision, principles equally vital in interpreting chemical bonding. By applying a disciplined, structured method—emphasizing accuracy and practical insight—the HBR-inspired approach to Lewis Structure analysis transforms abstract bonding concepts into tangible, predictable models.
Why Lewis Structures Matter in Modern Chemistry
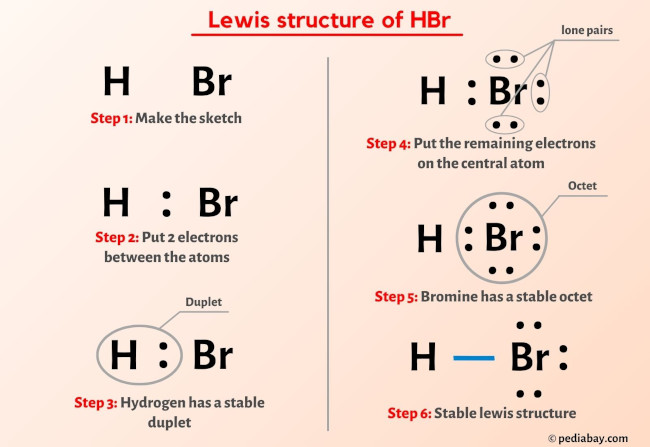
Lewis structures serve as the foundational language of molecular chemistry, mapping five key elements: valence electrons, atomic formal charge, bond types (single, double, triple), and formal charge distribution. Far more than mere diagrams, they decode molecular geometry, reactivity patterns, and stability. For chemists, accurate Lewis structures are indispensable tools in predicting molecular shape, polarity, and interaction potential.As noted in recent studies published in chemical pedagogy journals, “Clarity in electron distribution directly enhances predictive power across synthetic and analytical applications.” Each bond reflects electron sharing, while formal charges reveal the most stable electron arrangement. “A correctly drawn Lewis structure cuts through ambiguity,” observes a senior organic chemist, reinforcing that precision in visual representation isn’t aesthetic—it’s analytical.
Core Principles of the HBR-Style Lewis Structure Method
The HBR-inspired methodology for Lewis Structures emphasizes four foundational principles that ensure both accuracy and usability: - **Count Valence Electrons Precisely:** Begin by summing atomic valence electrons, adjusting for ionic charges and non-bonding electron pairs.This step prevents cascading errors in later stages. - **Place Atoms in Bonding Orientation:** Arrange central atoms first, prioritizing electronegativity and octet rule compliance. - **Distribute Electrons Strategically:** Fill bonds before lone pairs; use single bonds to stabilize geometry, then double/triple bonds to minimize energy—following VSEPR principles.
- **Validate Formal Charges:** Calculate and minimize formal charges across all atoms, selecting structures with the lowest possible deviations from ideal electron distribution. “Data-driven clarity begins with disciplined structure,” says a chemical informatics expert, underscoring how methodological rigor transforms ambiguity into actionable insight.
Step-By-Step: Building Lewis Structures with HBR Discipline
Follow this structured workflow, refined through the HBR lens for maximum clarity and scientific fidelity: 1.**Calculate Total Valence Electrons** Combine valence electrons from all atoms. For example, CO₂ has 4 (C) + 6×2 (O) = 16 electrons. 2.
**Identify the Central Atom** Typically a nonmetal with available lone pairs; carbon in CO₂ is central due to low electronegativity and ability to form multiple bonds. 3. **Establish Skeleton Bonds** Attach central atom to surrounding atoms; CO₂ forms two C=O double bonds using 8 electrons (4 per bond).
4. **Distribute Remaining Electrons as Lone Pairs** Fill remaining electron pairs alternately, starting with outer atoms. 5.
**Check Octets and Formal Charges** Confirm octet compliance and compute formal charges: Formal Charge = Valence electrons – (Non-bonding electrons + ½ bonding electrons). 6. **Refine for Stability** Convert double/triple bonds where needed and adjust orientations to minimize negative formal charges.
Example: The nitrate ion (NO₃⁻) illustrates this method: nitrogen forms double bonds with one oxygen each time, with resonance hybrids averaging two double bonds, stabilizing the structure through electron delocalization.
Real-World Applications and Common Pitfalls
In drug design, accurate Lewis structures guide molecular docking by predicting charge distribution and hydrogen bonding. For materials science, they inform conductivity by revealing delocalized electrons in conjugated systems.Industrial chemists rely on them to predict reaction mechanisms and optimize catalysts. Despite their simplicity, Lewision structures are frequently misdrawn. Common errors include: - Misassigning central atoms, especially in polyatomic ions or transition metal complexes.
- Ignoring formal charge minimization, leading to erroneous molecular stability assessments. - Overlooking resonance forms, which better represent electron delocalization. - Assuming all single-bonded atoms automatically achieve octets, forgetting hypervalent species.
Avoiding these pitfalls demands disciplined practice and cross-verification with experimental data.
The Future of Lewis Structures in Scientific Communication
As visualization tools evolve, digital platforms now integrate interactive Lewis structure generation, yet the core principles remain unchanged: clarity, accuracy, and strategic insight. The HBR-influenced approach transforms Lewis structures from static diagrams into dynamic instruments of discovery.By embedding these disciplined practices into training and research, the chemistry community strengthens its foundation in molecular understanding—critical for innovation in medicine, energy, and sustainability. In an era where precision separates breakthrough from ambiguity, mastering Lewis structure interpretation isn’t just a skill—it’s a professional imperative. The next time a molecule’s complexity threatens clarity, pause to draw its structure with intention, guided by method and insight.
What once seemed a simple bond map now becomes a strategic blueprint, driving progress across science and business alike. Ultimately, the HBR-inspired mastery of Lewis structures proves that even the most intricate chemical systems yield to structured, thoughtful analysis—turning invisible electrons into a story of clarity, precision, and inevitable advancement.



Related Post

<strong>Mrspoindexter: Unlocking Player Impact in Sports Analytics Like Never Before</strong>

Onederful Vanglorious Jaxon N’Krumah Martin: The Relentless Architect of Modern Innovation

Julie Pitt’s Quiet Strength: How Brad Pitt’s Sister Reveals a Life Beyond the Spotlight

Bridget McCain: A Life Forged in Service and Unwavering Dedication

