Unlocking Life's Secrets: Mastering DNA Extraction in Class 12 Biology
Unlocking Life's Secrets: Mastering DNA Extraction in Class 12 Biology
At the heart of genetics lies a molecule so fundamental yet remarkably manipulable—DNA. For Class 12 students diving into molecular biology, understanding how to extract DNA transforms abstract genetic concepts into tangible discovery. Far more than a classroom lab exercise, DNA extraction reveals the blueprint of life, empowering students to explore inheritance, identity, and the molecular basis of variation.
This guide unpacks the primary methods used in school laboratories, their scientific principles, comparative advantages, and practical nuances—equipping students to perform accurate, reproducible extractions that lay the foundation for deeper biochemical exploration. The journey begins with recognition that DNA, though pervasive in every cell, exists in trace amounts requiring careful isolation. Two principal approaches dominate educational settings: the phenol-chloroform method and the sodium acetate-ethanol precipitation technique—each with distinct procedural demands and educational value.
Understanding both enables students to appreciate methodology diversity, a cornerstone of scientific inquiry.
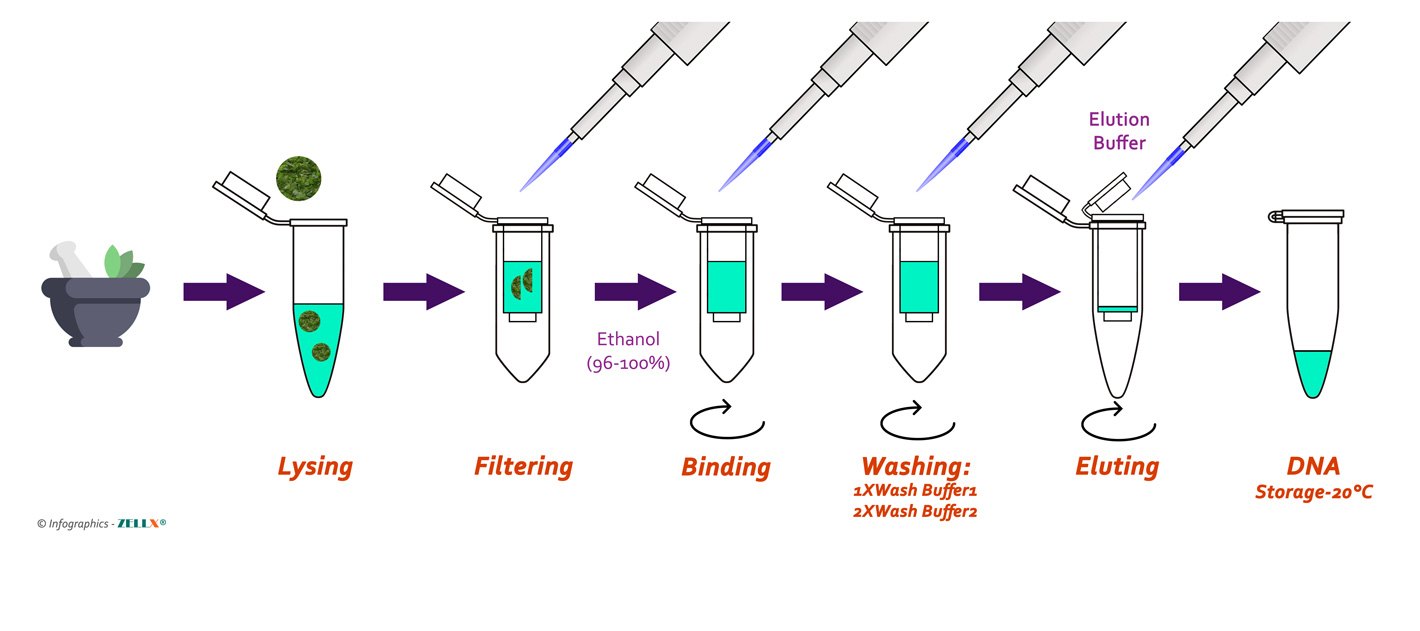
The phenol-chloroform method, long considered the gold standard for high-purity DNA isolation, relies on organic solvent extraction. In a typical procedure, crushed plant tissue or cell homogenate is mixed with phenol and chloroform, forming a two-phase system.
Proteins and lipids dissolve in the organic phase, while DNA remains in the aqueous upper layer—a cleavage exploiting differential solubility. Phenol’s denaturing effect also destroys nucleases, protecting DNA integrity. This method yields highly purified DNA, ideal for downstream applications like restriction enzyme digestion or PCR—yet demands strict handling due to phenol’s toxicity.
Students working in well-ventilated labs or supervised environments frequently apply this technique, gaining familiarity with sensitive biochemical workflows that mirror professional laboratory standards. In contrast, the sodium acetate-ethanol precipitation method offers a simpler, safer alternative increasingly adopted in well-equipped high school labs. This approach centers on denaturing DNA by altering solvent polarity: potassium acetate adjusts pH to destabilize DNA structure, while cold ethanol drives precipitation.
The process iteratively involves cooling homogenized samples, add of sodium acetate to precipitate nucleic acids, ethanol wash to remove salts, and final ethanol precipitation. The result is pellet-style DNA easily visible under a microscope—effective, cost-efficient, and accessible without hazardous chemicals, making it particularly suitable for classrooms emphasizing safety and scalability.
Regardless of method, successful DNA extraction hinges on several critical variables.
- **Sample Quality**: Fresh, intact tissue preserves superior DNA integrity; frozen or degraded samples often yield fragmented, low-yield DNA. - **Homogenization Efficiency**: Thorough grinding—using liquid nitrogen to freeze tissue—maximizes cell rupture, ensuring maximum DNA release. - **Contamination Control**: Minimizing exposure to RNAases via gloves, bleach-washed tools, and RNA-free reagents prevents unwanted nucleic acid degradation.
- **Timing and Refrigeration**: Rapid cooling halts enzymatic decay; ice baths and cold storage preserve sample stability before extraction. Students should also recognize that extraction success directly impacts downstream analyses. For instance, PCR amplification demands high molecular weight DNA to ensure proper product yield and specificity.
Similarly, agarose gel electrophoresis—often used to visualize extracted DNA—requires sufficient length and purity to produce clear, interpretable bands. A single oversight—like warm handling or inhibited incubations—can compromise results and undermine scientific conclusions.
Step-by-step execution of the sodium acetate-ethanol method illustrates the balance of simplicity and precision.
Begin by homogenizing 5–10 grams of plant material (such as spinach or strawberries) in a plastic tube with 1 mL of 10% sodium acetate solution. Add 2.5 mL cold 95% ethanol, gently mix by inversion, then chill ice bath for 30 minutes to allow precipitation. Centrifuge at 12,000 × g for 10–15 minutes to pellet DNA, carefully transfer the upper pellet to a new tube, and resuspend in 50–100 μL of 0.1 M TE buffer or water.
Successful extractions yield white-brown DNA pellets with characteristic clumping—evidence of extracted genetic material ready for genotyping or restriction analysis. Practice ensures reliability, particularly when avoiding rainbow loops or residual salt contamination that obscure visualization. Beyond hands-on lab work, understanding DNA extraction’s scientific underpinnings deepens conceptual mastery.
The process illuminates DNA’s double helix structure, nucleic acid solubility, and enzymatic sensitivity—all key to interpreting mutations, paternity tests, or forensic DNA profiling. Moreover, the principles apply across biotechnological frontiers: gene cloning, CRISPR editing, and personalized medicine all begin with purified genetic templates derived from such foundational techniques. Educators emphasize method transparency not only for safety but to foster scientific literacy.
When students compare phenol-chloroform yields with ethanol-precipitated DNA, they engage in critical evaluation: “Which method best preserves DNA integrity?” “How do reagent choices affect reproducibility?” These questions nurture analytical reasoning, mirroring real research practices where protocol selection shapes data quality and conclusions. In an age where genetic information permeates healthcare, agriculture, and criminal justice, demystifying DNA extraction equips Class 12 learners with both technical skill and ethical awareness. Handling this molecule demystifies life’s code—transforming curiosity into confidence and theory into tangible discovery.
As students master these methods, they step beyond the textbook into the very laboratory of innovation, ready to decode what lies at the core of all living systems.
Ultimately, DNA extraction in Class 12 is not merely a procedural exercise—it is the gateway to unlocking deeper biological truths. From phenol’s precision to ethanol’s simplicity, each method reflects a layer of scientific ingenuity, reinforcing the principle that even the most advanced biotechnologies begin with accessible, repeatable lab techniques.
For students poised at the intersection of education and discovery, mastering these extraction strategies ignites lifelong inquiry into the molecular language that defines life itself.



Related Post

Unlock the Future of Spatial Exploration with Pa Wmu Interactive Map

Joanne Cassidy-Tan: Architect of Visionary Tech Innovation and Inclusive Leadership

The Rise of Bolly4Uorg: Bridging India’s Love for Cinema and Global Access

Marilisa Maronesse: Decoding the Net Worth, Height, Nationality, and Age Behind the Public Persona

