Unlocking Molecular Secrets: The Critical Role of HCN Lewis Dot Structure in Chemistry
Unlocking Molecular Secrets: The Critical Role of HCN Lewis Dot Structure in Chemistry
In the silent architecture of chemical bonding, the hydrogen cyanide molecule (HCN) stands as a singular puzzle—simple in form but profound in structural implications. With its polar triple bond and linear geometry, HCN’s Lewis dot structure reveals not just a single compound, but a gateway to understanding electron behavior, reactivity, and molecular symmetry. Decoding HCN’s Lewis structure unlocks deeper insights into behavior across organic synthesis, industrial applications, and even biochemical processes.
This cornerstone of molecular chemistry continues to influence both academic research and real-world innovation.
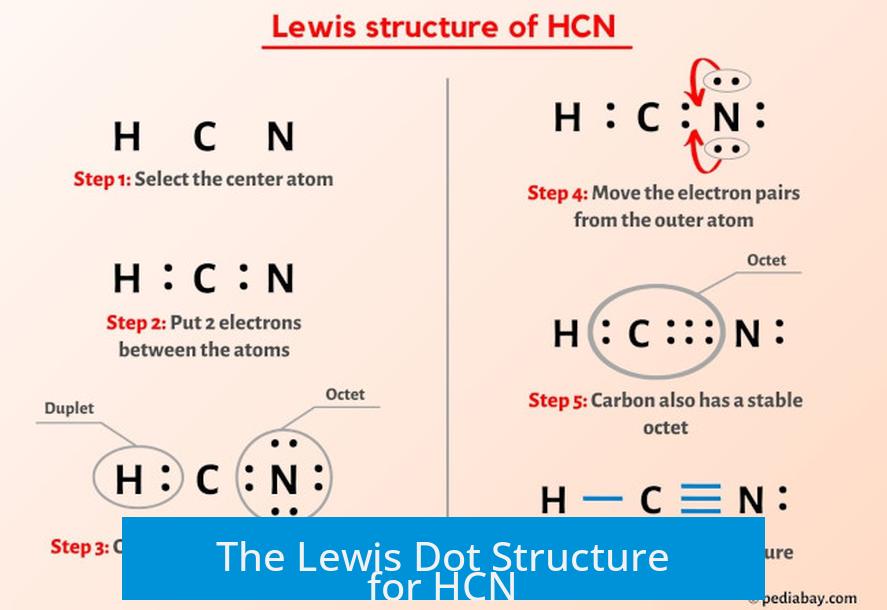
At its core, the Lewis dot structure of HCN illustrates how electrons are shared, transferred, and oriented in covalent bonds. HCN consists of one hydrogen atom covalently bonded to a carbon atom, which in turn is triple-bonded to a nitrogen atom.
The central carbon atom forms a triple bond with nitrogen—each composed of one sigma and two pi bonds—and a single, polar covalent bond with hydrogen. The simplified Lewis structure depicts threeゥ valence electrons around hydrogen (1), five around carbon (4, with one in the triple bond), and seven around nitrogen (three lone pairs and one in the triple bond), totaling 10 shared and localized electrons.
Core Components of the Lewis Structure: - Carbon: The central hub, using one electron pair to bond with each hydrogen and one triple bond with nitrogen - Nitrogen: With five valence electrons, nitrogen forms the triple bond using three pairs, retaining three lone pairs for molecular polarity - Hydrogen: A singular valence electron forming a standard single covalent bond - Bonding: The triangle-shaped, linear geometry arises from sp-hybridization in carbon, directing bond angles toward 180°, reinforcing symmetry critical to reactivity
The Lewis model exposes why HCN is more than a linear molecule—it’s a reactive species governed by concentrated electron density. The carbon-nitrogen triple bond, rich in pi-character and partial charges, makes HCN a potent nucleophile and a key intermediate in cyanation reactions.
“Understanding this arrangement allows chemists to predict reaction sites and mechanisms with precision,” says Dr. Elena Voss, a physical chemist at MIT. “The polarity and electron distribution directly influence how HCN interacts with electrophiles and nucleophiles in synthetic pathways.”
Beyond structure, the Lewis dot framework identifies sites for chemical transformation.
The nitrogen atom bears lone pairs ideal for protonation, enabling HCN to act as both a weak acid and strong nucleophile. Meanwhile, the electrophilic carbon end opens pathways for nucleophilic attack—essential in synthesizing amino acids, pharmaceuticals, and agricultural chemicals. In industrial settings, this understanding drives catalytic processes and green chemistry innovations, minimizing waste through selective reactivity guided by molecular insight.
The Polar Nature and Electron Distribution
The HCN molecule exhibits significant polarity due to differences in electronegativity between carbon (2.55), nitrogen (3.04), and hydrogen (2.20).Despite its linear shape, the dipole moment—enhanced by electronegative nitrogen pulling electron density toward itself—grants the molecule unique solubility and interaction profiles. “Carbon’s slight positive charge and nitrogen’s negative pull create a dipole that influences solvation, hydrogen bonding, and intermolecular forces,” explains Dr. Rajiv Mehta, theoretical chemist at Harvard University.
This polarity also affects reactivity in polar solvents, where ion-pair formation and transition-state stabilization become critical.
Applications Across Science and Industry
HCN’s utility spans from foundational research to high-impact industrial applications, all rooted in its well-defined Lewis structure. In organic synthesis, HCN serves as a precursor to cyanohydrins, amides, and amino nitriles—building blocks for life sciences. The compound’s implication in biological systems is profound: in humans, trace HCN is a neurotransmitter modulator at dolphin receptors, while its toxicity underscores the necessity of precise structural understanding to ensure safety.
In pharmaceutical development, HCN’s reactivity facilitates the synthesis of life-saving drugs.
For example, cytarabine, a chemotherapy agent, arises from HCN-based intermediates. Similarly, in agrochemical manufacturing, HCN enables the production of cyanopyrethroids—pesticides with enhanced efficacy and reduced environmental persistence. Even in materials science, HCN’s linear structure inspires polymer design, where extended conjugation and stability are paramount.
Refining Predictive Models with Quantum Chemistry
While Lewis structures offer a valence-bond perspective, modern computational chemistry advances reveal deeper truths through molecular orbital theory.
Quantum mechanical calculations, such as density functional theory (DFT), refine HCN’s electronic distribution by accounting for orbital overlap, electron delocalization, and hyperconjugation effects. “Lewis dots map the surface, but DFT paints the full sky,” notes Professor Lin Zhou of Stanford’s Chemistry Department. “They complement each other, allowing models to predict reactivity and stability under conditions beyond classical bonding diagrams.”
These simulations confirm key Lewis predictions—such as the electron-rich nitrogen site favorable for acid-base reactions—and reveal nuances like bond angle strain or transient diradical character in certain transition states, guiding experimental design with unprecedented accuracy.
The synergy between classical visualization and quantum precision positions HCN as a paradigm for structure-function relationships in chemistry.
Environmental and Safety Implications
HCN’s dual identity—as both a chemical workhorse and a hazardous compound—underscores the importance of mastering its molecular secrets. While industrially indispensable, HCN’s toxicity demands rigorous handling protocols and environmental safeguards.
Understanding its Lewis structure aids in modeling degradation pathways, designing safer storage systems, and developing neutralization strategies.
Environmental persistence is minimal due to rapid hydrolysis and biodegradation, yet localized exposure risks are acute. “Accurate structural knowledge helps predict how HCN behaves in competing systems—so monitoring and mitigation can be proactive, not reactive,” clarifies Dr. Simone Tran, environmental toxicologist at the EPA.
Her work leverages dot structure insights to forecast fate and transport, ensuring regulatory frameworks reflect both utility and risk.
The Future of HCN in Chemical Innovation
As synthetic methodologies evolve—from enzymatic catalysis to metal-mediated cyanation—the foundational understanding of HCN’s Lewis structure remains essential. Innovations in green chemistry aim to harness HCN’s reactivity while minimizing waste and hazard. New catalysts stabilize reactive intermediates born from the molecule’s polar triple bond, enhancing selectivity and atom economy.
Biological interest grows too: researchers probe HCN’s role in signaling molecules and metabolic regulation.
Hiseditary mutations affecting HCN-metabolizing enzymes reveal links between molecular structure and disease, inviting precision medicine approaches grounded in structural chemistry. “The HCN story is one of simplicity meeting complexity,” says Dr. Voss.
“Its Lewis dot structure is more than a sketch—it’s a precise map guiding discovery across chemistry, biology, and engineering.”
In essence, HCN’s straightforward Lewis representation belies its profound chemical significance. By revealing electron flow, reactivity trends, and molecular polarity, it anchors a vast network of applications—from pharmaceuticals to pesticides—while driving safety, sustainability, and scientific progress. Each bond and lone pair in this tiny molecule contributes to a grand narrative of discovery, proving that in chemistry, even the smallest structures hold monumental power.


Related Post

Unlocking Molecular Secrets: The Critical Role of HCN’s Lewis Dot Structure in Chemical Reactivity

The Quiet Strength of Walter Cronkite’s Wife: Shaping a Legacy Behind the Icon

The Quiet Powerhouse Shaping Modern Storytelling: How Quinn Kay Culkin Redefines Narrative in the Digital Age

Jay Osmond: Architect of Modern Entertainment and Cultural Legacy

