What Are The Charges Of A Proton? The Charged Core That Shapes Matter
What Are The Charges Of A Proton? The Charged Core That Shapes Matter
At the heart of every atom lies a particle of profound significance: the proton—a positively charged subatomic particle that defines the structure and behavior of matter across the universe. With a charge of exactly +1 elementary charge, the proton is not merely a structural component; it is the key determinant of chemical interactions, atomic stability, and the very nature of the periodic table. Understanding what a proton’s charge truly means reveals fundamental truths about electromagnetism, quantum mechanics, and the forces that bind the cosmos together.
Each proton carries a well-defined electrical charge equal to +1.602 × 10⁻¹⁹ coulombs, a value approved by generations of experimental physics. This charge is both intrinsic and universal—identical in every proton, regardless of its environment. In practical terms, this means that protons contribute positively to the electric charge of an atom, balancing negatively charged electrons and defining the element’s identity.
But the charge of a proton is far more than a simple number—it is the foundation of atomic chemistry. “The charge of the proton is the cornerstone of atomic structure,” explains Dr. Elena Marquez, a physicist specializing in particle physics at CERN.
“Without this fixed positive charge, electrons would not be bound to nuclei, and elements as we know them could not exist.”
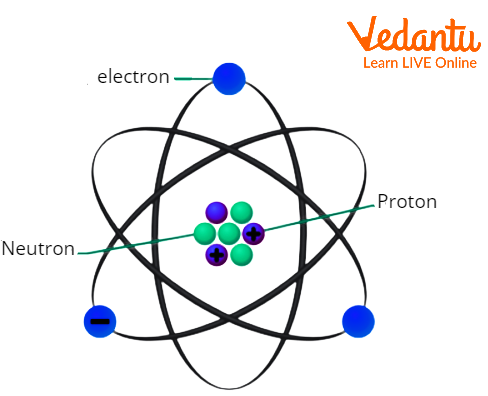
Protons, together with neutrons, constitute the atomic nucleus, with protons determining the atomic number and thus the element’s place in the periodic table. An atom of hydrogen contains one proton; carbon has six; iron features 26. The numerical value of this atomic number stems directly from the number of positively charged protons in the nucleus.
For example, a neutral carbon atom has six protons, meaning its electric charge sum exactly equals +6e—while neutrality elsewhere arises from balanced electrons. What gives a proton its charge? Protons are composed of three "quarks"—two up quarks (each with +2⁄3 e charge) and one down quark (with –1⁄3 e charge). The mathematical sum of these quark charges produces the proton’s overall +1 charge: (2⁄3) + (2⁄3) + (–1⁄3) = +1 This internal architecture explains why the proton’s charge remains exact and invariant.
It is not borrowed or influenced by external fields; rather, it is embedded in the quark-gluon framework of quantum chromodynamics.
Charge regulation is fundamental. Unlike neutrons, which are electrically neutral, protons carry a stable +1e charge at all known scales.
Measurements using ultra-precise Penning traps confirm this value to within one part per billion. Such precision underscores both the consistency of physical laws and the role of the proton as a reliable atomic unit. Why does proton charge matter beyond atoms? The positive charge governs electromagnetic forces, influencing everything from chemical bonding to material conductivity.
“In chemistry, the proton charge dictates electron behavior—reactions occur because of how positively charged nuclei attract electrons,” notes Dr. Marcus Reed, a materials scientist at MIT. “This charge governs periodic trends, reactivity, and bonding types.”
In nuclear physics, proton charge also plays a pivotal role.
Stable isotopes maintain balance between positively charged protons and electrically neutral neutrons—whenever proton count increases, neutron count must rise to counteract growing repulsion. Deviations disrupt atomic stability, leading to radioactive decay. “Charge imbalance is mother nature’s warning sign,” says Dr.
Marquez. “A surplus or deficit in protons triggers decay pathways, shaping the life cycle of elements.”
Size and scale: how does the charge manifest? Though the proton is tiny—about 1 femtometer (10⁻¹⁵ m) in diameter—its charge radiates across vast distances due to Coulomb forces. This influence extends beyond individual atoms: in solids, liquids, and plasmas, proton charges underlie conductivity, polarization, and phase behavior.
Even in cosmological contexts, proton charge affects particle interactions in stellar interiors and interstellar media.
Experimental verification and ongoing research Proton charge has been measured and verified for nearly a century, using techniques ranging from mass spectrometry to quantum electrodynamics calculations. The CODATA diagonalization group maintains the accepted value, which serves as a cornerstone in international metrology.
Yet, physicists continue exploring whether this charge could vary across space or time—a question that, if proven false, reinforces the stability of physical constants essential to cosmology and fundamental theory. The proton’s charge in perspective To summarize, the proton’s charge is not just a unit of measurement—it is a fundamental constant that anchors atomic physics, chemistry, and nuclear science. Defined with extraordinary precision, it ensures consistency across disciplines and civilizations.
From subatomic force carriers to macroscopic materials, the proton’s +1 charge governs interactions, balances energy, and defines identity. It is, quite simply, the electrified key to the structure of matter.
As science advances, the proton’s charge remains unchanged—its constancy a silent assurance of the predictable laws that govern our universe.
Understanding this charge deepens our grasp of reality itself, revealing how a single, positively charged particle holds the cosmos in precise electrical balance.



Related Post

Wife Of Howie Long: A Life Beyond the Spotlight, Anchored in Love and Resilience

Mastering Grade 7 Math: 40 Essential Practice Problems That Shape Senior Learning

Victoria Spader Is the Only Woman James: Redefining Legacy in Entertainment

Ames Mcnamara Wiki

