What Is the Protein Monomer? The Tiny Building Blocks That Shape Life
What Is the Protein Monomer? The Tiny Building Blocks That Shape Life
Proteins are the invisible architects of biology—complex, dynamic structures that govern nearly every process within living cells. At the heart of this intricate architecture lies the protein monomer, the fundamental unit responsible for life’s molecular construction. These microscopic building blocks, though invisible to the naked eye, orchestrate functions ranging from cellular transport to enzymatic catalysis, forming the backbone of every living organism.
Understanding the protein monomer is essential to grasping how life, at its most basic level, is constructed from simple yet sophisticated components.
Every protein is assembled from one or more protein monomers—repeated units of amino acids linked through peptide bonds. “Amino acids are the alphabet of life,” explains Dr.
Elena Marquez, a structural biologist at the Institute for Biochemistry. “When they join in specific sequences, they fold into functional proteins capable of performing extraordinary tasks.” Each monomer consists of a chain of 20 naturally occurring amino acids, selected from a standardized genetic code. This sequence determines how the protein folds, and folding dictates function.
As Dr. Marquez notes, “The monomeric structure is not just a start point—it’s the foundation upon which biological purpose is built.”
The protein monomer’s significance lies in its role as the repeat unit in larger protein architectures. Polymers such as collagen, keratin, and hemoglobin are built from countless linked monomers arranged in precise patterns.
Collagen, the most abundant protein in mammals, relies on triple-stranded helices formed by three coiled monomer chains—each composed of glycine, proline, and hydroxyproline., enabling tendons, skin, and bones to maintain structural integrity.
Each amino acid within a monomer contributes distinct chemical properties crucial to protein stability and activity. The side chains—ranging from nonpolar and neutral to positively or negatively charged—dictate how monomers interact with one another and their environment. Hydrophobic amino acids cluster inward in the protein core, shielding themselves from water, while hydrophilic residues face outward, engaging in functional interactions.
This selective arrangement drives the spontaneous folding process central to protein function.
Monomer formation begins during translation, when ribosomes string together amino acids into precursor chains based on messenger RNA instructions. Once synthesized, these nascent chains undergo folding, cleavage, and sometimes post-translational modifications to yield mature, functional proteins. The process is guided by molecular chaperones—proteins that assist proper folding—ensuring that monomers assume correct three-dimensional conformations essential for activity.
“Improper folding turns a promising monomer into biological static,” warns Dr. Rajiv Nathan, a biophysicist at Stanford University. “Even a single error in monomer assembly can disrupt entire cellular machinery.”
The versatility of protein monomers enables a staggering diversity of biological functions.
Enzymes, for instance, deploy specialized monomers to accelerate chemical reactions with remarkable precision. Antibodies rely on monomeric immunoglobulin units that bind pathogens with high specificity. Hormones like insulin form multimers—though composed of repeating functional monomers—to regulate metabolism across the body.
Each monomer, though simple in form, contributes to a collective intelligence that sustains life’s complexity.
Modern research continues to reveal deeper layers of monomer behavior. Advanced techniques such as cryo-electron microscopy and X-ray crystallography expose atomic-level details of monomer interactions, enabling scientists to design synthetic proteins with novel functions.
In medicine, this knowledge fuels innovations in targeted drug delivery, biomaterials, and regenerative therapies—all anchored in the monomer’s core principle: simplicity breeds function, and specificity defines life.
The protein monomer is more than a biochemical unit—it is the cornerstone of biological architecture, a microscopic marvel whose repetitive elegance underpins the complexity of every organism. From microscopic enzymes to macroscopic organs, life’s story is written in these tiny, linked building blocks. Understanding their role not only illuminates fundamental biology but opens pathways to transformative scientific progress.
As research advances, the protein monomer remains a central figure in the ongoing exploration of life’s molecular blueprints.
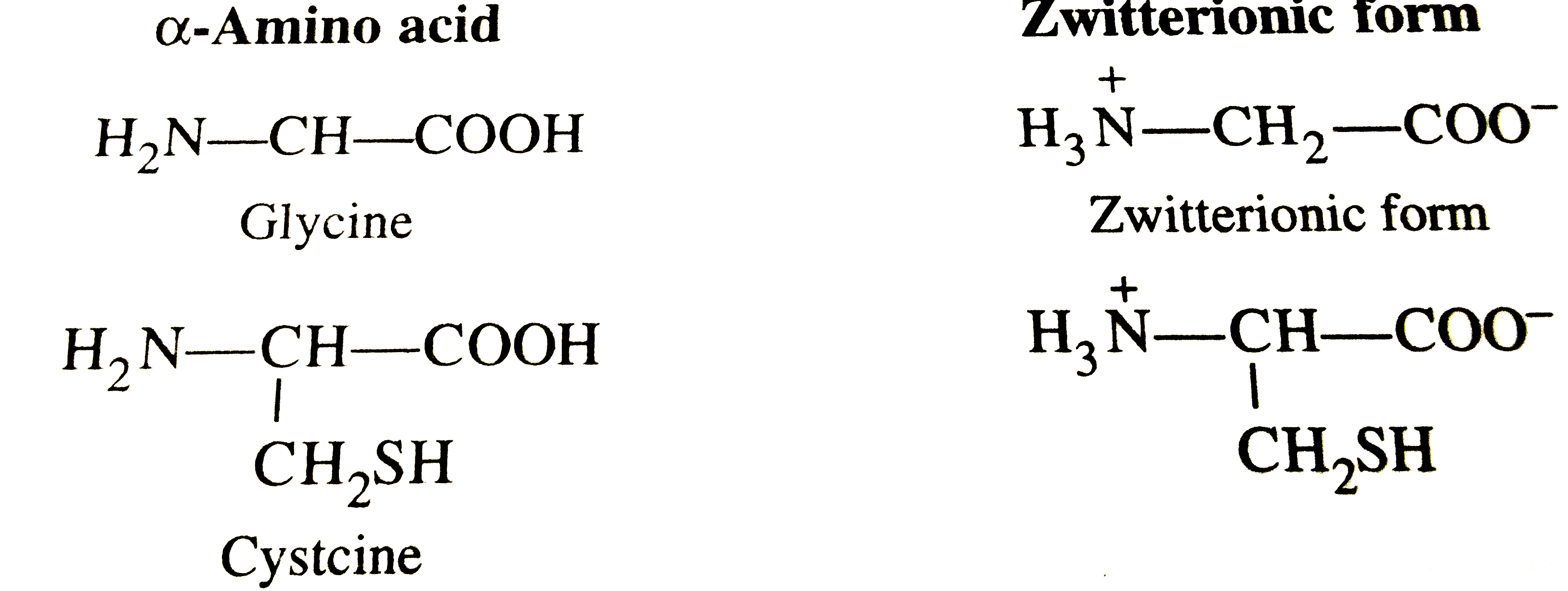


Related Post

Asia Kate Dillon: Redefining Visibility, Identity, and Representation in Modern Media
Brandon Peniche: Architecting Authenticity in a Digital-Driven World

Ted Nugent Band: The Raw Power of Country Rock’s Relentless Elevation

The Unscripted Chemistry: Decoding the Dynamic Between Ms Niko and T.I.

